We can’t stress enough the damage that can be caused to old soft tombstones and monuments, when harsh chemicals and procedures are applied. We have given you the shortest and simplest means to damage free cleaning under BASIC STANDARDS…Cleaning. That list is quite short. The long list consists of the many bad things that are used and have been used over the years for cleaning. We will try to list as many of these as we can and focus on the most prevalent culprits. We will also give the best explanations science and studies have to offer on this subject. Remember: anything other than what we have as an approved cleaner, will do some degree of damage. It’s merely a matter of what degree of damage. Stone is much like wood when it comes to taking in moisture. Softer stones can be like giant sponges and that is the biggest danger and concern over what to apply on them. Common everyday soaps to harsh chemical will travel deep into the stones and cause damage to any type of custom stonework from the inside out. This is the type of damage that can’t be detected until it’s too late.

Harmful Cleaners and Cleaning Methods
When it comes to tombstone and monument cleaning, the most important thing about any liquid cleaning solution is ph neutral.

Bleach is at the top of the list as the number one harmful cleaner most used. Most who use it do because it’s cheap, kills germs and fungi, and it will bleach white a marble stone. Sounds good in theory, but very harmful. The makeup of bleach – The chemical compound formula for sodium hypochlorite is NaOCl. – reaction – Chlorine and sodium hydroxide will produce sodium hypochlorite Cl2 + 2NaOH = NaOCl + NaCl + H2O. Bleach is damaging do its sodium content. The damaging salt deposits left behind in the stone will break down the stones molecular structure from within.


Bleach quite often has an accomplice with equally devastating effects, known as a wire brush.
A wire brush will do extreme damage by removing layers of stone. Sometimes removing entire inscriptions. It becomes compounded damage when used with bleach. Please save the wire brush for your next auto body restoration, not your next trip to the cemetery.
ALL typical household cleaners contain some amount of cleaning agent or byproduct such as sodium that will do some degree of damage. These products are best left under the kitchen sink or in the garage where they can be used around the home and on what they were intended for.

Cleaning with power washers is also not recommended. High pressure water has been known to take layers of skin off of a person. If you can strip paint off of a building, you can strip layers of stone off of a grave marker. Many historic stone structures have had their historic mortars and soft bricks damaged with this process. The two key factors here are, the amount of pressure and the softness of the stone. The real damage occurs when overzealous people turn the pressure up on soft stonework.

This process should only be managed by professionals who use it on a limited basis if at all. Many preservationists want nothing to do with it what so ever and others use it sparingly. But since some historical professionals and historical institutions use this technique, it would be disingenuous of us to not try and represent this subject equally. We caution against it because typically there are too many factors involved and too many things can go wrong very easily. This coupled with the factors of equipment and the large water supply needed, most find it not practical.
Sandblasting on the other hand should never be used as a method of cleaning. The main point of sandblasting is to remove unwanted material to produce a new prep surface. This means historic stone material will be removed to achieve this. Removing historic stone material from the surface will destroy a stones natural protective skin and cause the stone great harm over a short time.


ANY cleaning of a tombstone or monument by mechanical means is devastating to the stone and not recommended. Stone cleaning is meant to be done with the least invasive methods. Mechanical cleaning is the opposite of that. It is very abrasive and extremely invasive.
As we have stated before, stones have a molecular structure and are complex miniature microcosm worlds of nature. And like most things in nature, they change and develop defense mechanisms. When a stone is below ground in a quarry, it has a different molecular quality than one from above ground. A virgin type quality if you will that has not been exposed to both natural and man-made elements. It’s busy being part of the ever changing waxing and waning processes that take place below the earth’s surface. When it is quarried and brought to the surface, it stands bare and naked to the torchers of the world’s atmosphere. And in response to this, it grows a protective skin in an effort to shield itself from these elements
When you use an abrasive means to clean a stone, you strip away this cloak of protection and leave the stone totally defenseless. This is much like cleaning your teeth with a dremel and removing the protective enamel coating.

Ken Follett – founding member and first president of the Preservation Trades Network, experts in the field of historic preservation, was asked the following question.
What are some of the harmful effects that result from using high speed rotary nylon wheels on power tools on gravestones?
“I am a solid advocate of D/2 biological solution. That regardless, the technique of treatment of stone surfaces of cultural heritage value. Polishing with Nyalox brushes on a power drill is totally off the charts on an international basis in the world of stone and monument conservation. Likewise, the bad thinking that gravestones need to be, “returned to how they looked originally”. The very first questions needs to be, “Why do this at all? What is the necessity? Where in our culture does this need come from?”

A quote from Lynette Strangstad, Author of “A Graveyard Preservation Primer” The premier book on cemetery conservatorship.
“Briefly in my opinion, “polishing” an old gravestone is not appropriate. The entire stone is altered. Some of the surface is removed. And that fragile surface is the very reason most consider the stone valuable. (Though that is only part of the significance) In grinding the surface, that is polishing, one is removing part of the lettering. Three or four such abrasive cleanings over time, say 15 or 20 years, could easily equal the stone loss that would occur naturally in a 100 or more years. It’s good to remember that care for gravestones is not just to satisfy our aesthetic desire in the present; it is to preserve the stone for future generations. D/2 is an effective and responsible cleaning agent when needed”.
People who use this method attack a stone in a series using all three of these wheels. Note the claim to be far superior to wire brushes lasting 10 X as long. These are aggressive tools used in auto body prep work.
Ken Follett and Lynette Strangstad are two of the most knowledgeable and respected people in this field and have years of studies and experience on their side. They are a prime example of the experts CCUS members gain their knowledge from.

The Midwest has been in the grips of a mechanical cleaning craze for roughly the last 10 years. Our CCUS Colorado member Dianne Hartshorn, can attest that it has traveled from Indiana to Colorado. Several Indiana based companies practice this destructive method and one continues to teach it. This has become known as the “Nylon Drill Cleaning” method. This method used to have a much larger stage that it operated from. But due to many voices coming out in opposition, it has declined and moved underground.

People who attend these classes are completely unaware of the damage that is caused by this method. This is very understandable when an individual or company claims to be an expert in this field. This becomes an even greater problem when others perpetuate this situation over and over, and tout it as the greatest thing since baked bread where grave marker cleaning is concerned. This is how urban legends are born.
Dispelling damaging methods like these and shedding light on no harm methods is a big reason we formed the CCUS. One of our goals was to provide you a place to thoroughly vet preservationists and preservation methods. And thus giving you a group of professionals to choose from to teach a class, perform work, or be advised by.
Why removing the outer layer of a grave marker is a very damaging thing
An article by Geologist Don Hilton
All rock begins to weather from the moment it is removed from the earth. It doesn’t matter what kind of rock it is, or how tough it is, it begins to weather.
It’s easy to find grave markers heavily damaged by the environment. Low-quality marble is regularly mentioned as a stone type that is quickly ruined by exposure to acid precipitation. Alternatives are just as easily found: Sandstone that spalls due to water infiltration or slate that splits because of uneven heating from the sun. But something else is going on beside the obvious damage that we can see with our eyes.
Imagine a loaf of freshly-baked bread. Why does it have a crunchy crust? Really… the whole loaf was in the oven. There was nothing special about the outside layers of the dough. Why is the outside of the bread so different than the inside?
You know the answer. The outside was exposed to the heat and dryness of the oven. As the crust formed, the inside was protected from the worst of the extreme environment. In a properly baked loaf, the hard crust and soft bread are easy to tell from one another. They are made up of the same stuff, but different due to environmental exposure. They are all part of one thing and, while attached, they can be pulled apart, one from the other.


The rock that makes up a grave marker is the same way. When it’s pulled out of the ground, it’s all the same thing. The face of a commercial stone is worked: honed, polished, and carved. Then, it’s stuck out in the open, just like kneaded bread dough is put in an oven.
Just like bread dough in a hot oven, being exposed to the environment changes the surface of the rock. Sometimes the change is rapid, like a dissolving marble. Sometimes the change is slow. Sometimes you can’t even tell if it’s taking place, but it is. In fact, you can bet on it.
What happens depends on the kind of stone. The surface may become soft and brittle, or it may develop a protective “skin” or patina. These changes are the result of the rock trying to come to a physical and chemical equilibrium with its environment. Like baking bread that forms a crust.
The white marble stone to the left carved in 1877, is showing some signs of dissolving with its natural exposure to the environment.
These altered, outer rock surfaces are easily damaged by abrasive mechanical or improper chemical cleaning. When created, grave markers are frequently “dressed” with tools to give them a specific texture that contributes to their historic character as much as any ornately carved stonework. Such detail is damaged by harsh cleaning techniques, either abrasive and/or chemical. The pattern of tooling or cutting is erased, and the crisp lines of carving are worn. Aggressive cleaning removes the polished or honed surface of marble or granite, almost like your “frosting” a clear glass.


The white marble stone above was mechanically cleaned with what is known as a “Nyalox” wheel used on the end of a drill. These wheels are advertised as “abrasive impregnated nylon brushes” with bits of metal that are supposed to last 10 times longer than a wire brush. This stone, once very legible before this mechanical cleaning, is an example of what happens when the protective skin/crust of a stone is removed by harsh or abrasive means, as stated below. This accelerated deterioration took under 4 years to happen.
A cleaning procedure that is too strong removes the protective “crust” or “skin” of the stone. This exposes a new and increased surface area to collect moisture, dirt, and bio-growth. This accelerates weathering. This speeds damage.
Think about it… If halfway through the baking process, what would happen if you pulled the bread from the oven and peeled away the crust? Would you end up with bread without a crust? Nope. You’d end up with a ruined loaf of bread.
And that’s why you don’t want to clean a grave marker too harshly and chance removing any of its surfaces. You’re not making it cleaner. You’re not making it better. You’re ruining it.
Ammonia…aggressive cleanings other half of the 1, 2, punch that damages grave markers
An article by Geologist Don Hilton and Cemetery Conservator Mark Morton
For years ammonia has been viewed as a safe and inexpensive cleaner for grave markers. And the golden rule has always been to use it at a ratio of 5 parts water to 1 part ammonia. And to never go above that ratio by increasing the amount of ammonia. Ammonia on its own is very alkaline on the pH scale, coming in at 11.6. To at least begin to understand the effects of ammonia that we are describing, we need to look at some basic chemistry and have a basic understanding of the importance of pH and pH neutral. The scale and description below should do just that.
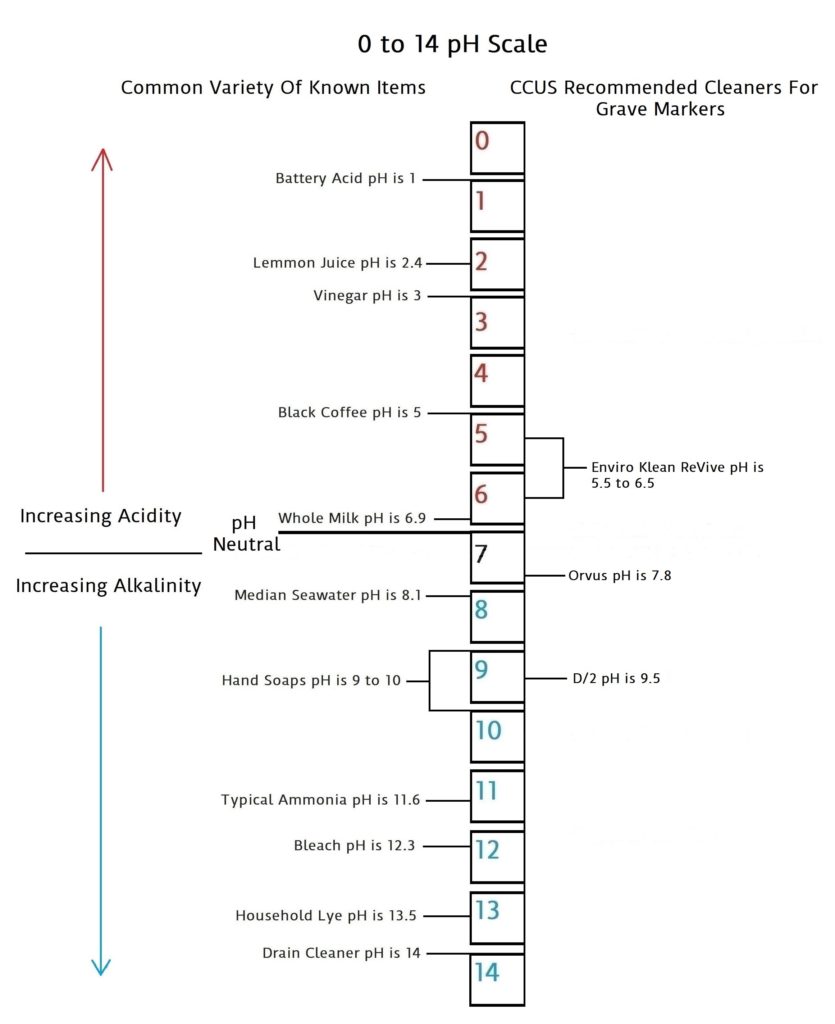
What Is pH And Why Is It Important?
pH is a scale of 0 to 14 that measures acidic quantities and qualities against alkaline quantities and qualities. A pH less than 7 is said to be acidic and solutions with a pH greater than 7 are basic or alkaline. pH neutral is 7. Human pH neutral is 7.30 to 7.45 slightly alkaline. Being as close to pH neutral is important because this is the least caustic area of the scale between the two extremes.
This scale will give you an idea of how pH is measured using common items you may be familiar with.
Acidic and basic are two extremes that describe a chemical property of chemicals. Mixing acids and bases can cancel out or neutralize their extreme effects. A substance that is neither acidic nor basic is neutral.
The pH scale measures how acidic or basic a substance is. The pH scale ranges from 0 to 14. A pH of 7 is neutral. A pH less than 7 is acidic. A pH greater than 7 is basic.
The pH scale is logarithmic and as a result, each whole pH value below 7 is ten times more acidic than the next higher value. For example, pH 4 is ten times more acidic than pH 5 and 100 times (10 times 10) more acidic than pH 6. The same holds true for pH values above 7, each of which is ten times more alkaline (another way to say basic) than the next lower whole value. For example, pH 10 is ten times more alkaline than pH 9 and 100 times (10 times 10) more alkaline than pH 8.
Pure water is neutral. But when chemicals are mixed with water, the mixture can become either acidic or basic. Examples of acidic substances are vinegar and lemon juice. Lye, milk of magnesia, and ammonia are examples of basic substances.
Ammonia as an industrial fertilizer
Ammonia (NH3) is the foundation for the nitrogen (N) fertilizer industry. It can be directly applied to soil as a plant nutrient or converted into a variety of common N fertilizers, but this requires special safety and management precautions. Ammonia has the highest N content of any commercial fertilizer, making it a popular source of N despite the potential hazard it poses and the safety practices required to use it. For example, when NH3 fertilizer is applied directly to soil, it’s in a pressurized liquid that will immediately become vapor if exposed to air after leaving the tank. To prevent such releases into the atmosphere, growers use various tractor-drawn knives and shanks to place it at least 10 to 20 cm (4 to 8 inches) below the soil surface. Ammonia will then rapidly react with soil water to form ammonium (NH4+), which is retained on the soil cation exchange sites.
Ammonia as a simple fertilizer by adding water
Ammonia is sometimes dissolved in water to produce aqua ammonia, a popular liquid N fertilizer. Aqua ammonia doesn’t need to be injected as deeply as NH3, which provides benefits during field application and has fewer safety considerations. Aqua ammonia is frequently added to irrigation water and used in flooded soil conditions.
Summation: When you take household ammonia and add water, you are making a form of “liquid N fertilizer”. And even though this type of fertilizer does not need to be “injected deeply”, the porosity of stones like marble, sandstone, limestones, etc. will in a way, self-inject this like a sponge due to the stones natural porosity. Most all cleaning that occurs on grave markers is to remove or kill biological growth such as lichens and moss. Many of these biological growths attach themselves deep into the stone, making them very difficult to dislodge. So when you apply your homemade liquid N fertilizer in an effort to get rid of biological growth, you are actually fertilizing it at its core where it resides below the stones surface. If the stone already has a “protective crust or skin” as described above, it at least has some natural protection to begin with. But when you mechanically remove that layer as you spray down the stone with your water and ammonia solution, you are laying the stone bare to everything and greatly aiding by injecting this into the stone. This is why we refer to this as a 1, 2, punch that damages a porous stone forever.



Above left to right, are 3 white marble stones that were cleaned with the Nyalox wheel and ammonia. The first stone was already showing signs of natural dissolving, or as many conservators call it, “the melted candle wax effect”. The middle stone represents the average amount of damage that occurs. The stone on the right is an example of how the Nyalox wheel has ground off most of the delicate detailed carving, and is beginning to show some biological regrowth. All 3 of these stones were completely white directly after the Nyalox method was completed. You never know for sure the severity of damage that will occur. Cloverdale Cemetery, Cloverdale Ind.
In addition to this, you have opened up the stone to a plethora of elements natural & manmade. Let’s start with the natural elements. We have all of the biological growths that occur naturally in nature as the natural process that breaks down everything on earth. This is further accelerated as wind and rain leave soil deposits as part of this cycle. So you roughly have soil and seeds and seasonal growth patterns. Manmade elements can range from a wide variety of atmospheric pollutants and acid rains, to any sort of carry over used in the farming industry, from fertilizers to pesticides. So the grave marker becomes very much like a petri dish.
Preserving anything is always a fight against natural occurrences in nature. And the best way to combat this when it comes to grave markers is with the least invasive methods possible. It’s a matter of not going too far in an effort to make something look brand new, but to make a “do no harm” improvement. And realizing where the tipping point is between the two. And also realizing when something is as good as it will get or to leave it alone altogether.
SOURCES:
https://www.britannica.com/science/desert-varnish
https://pubs.usgs.gov/pp/1210/report.pdf
https://www.thoughtco.com/chemical-weathering-p2-4122736
Photo Courtesy…Mark Morton and Lee Creed, Images by Cemetery Conservators For United Standards

There has been much said, both negative and positive about this product for grave marker cleaning since it first came onto the scene a few years ago. This product has been quite popular among those looking for the quick, cheap, and easy to find solution for cleaning biological growth off of gravestones. Those in favor of this product site the previous reasons and back that up with the manufacturer’s claims.
Those not in favor of this product point out reasons not associated with easy button reasons of convenience and manufacturer’s claims. Their reasons are about being fact based due to rigorous testing and analysis in accordance with the “do no harm” practices for cemetery preservation and restoration. Their concerns are as follows.
Concern over this product having strong diverse chemical combinations that “may” damage old porous stones such as marble, sandstone, and other siltstones.
Concerns about an MSDS sheet with many warnings about safety precautions that have fluctuated over the past several years.
A complete lack of any official independent lab or field study showing the products chemical makeup or longer term effect on historic stone.
And some voice concerns about a product they fear may have some sort of sealant quality, in their eyes, because many find that water beads on the stone after this product is applied. And there for, they worry the product is interfering with wicking, water travel, and natural water displacement in and out of the stone.
The do no harm practice view is all about the use of products that have been tested and proven, as much as is possible, to best insure damage does not occur if used on historic stone. Wet & Forget has no such study at this time that we are aware of. On the positive side, this product does have several similar qualities in its chemical makeup that other “approved” biocide products have. But, without proper analyzation, it is hard to tell just how these chemical combinations actually interact with one another. This product should be analyzed and rigorously tested in much the same manner as D/2 Biological Solution was tested, to best insure it does not do harm. And so, we can neither condemn nor condone this product until such testing has been done. As always…we caution you not to use untested products, and to please use proven do no harm products. Why take chances?
The information about Wet & Forget can also be found HERE.